Nano experiment kit for Chemistry
Why does the characteristic colour of gold change when its particles are smaller than 5 nm? Where can such effects be applied in industry today? How can lycopodium be super-hydrophobic and also cause powerful explosions?
This experiment kit provides relevant answers to all those questions. We hope that the experiments described here will help you pass a few interesting hours in the world of small
particles and fine structures.
Some of the experiments introduced here are intended for demonstration only, but others can be incorporated into lessons under the supervision of a teacher without excessive preparation.
We hope that through our experiment kit we can bring the fascinating world of chemistry closer to your pupils and you.
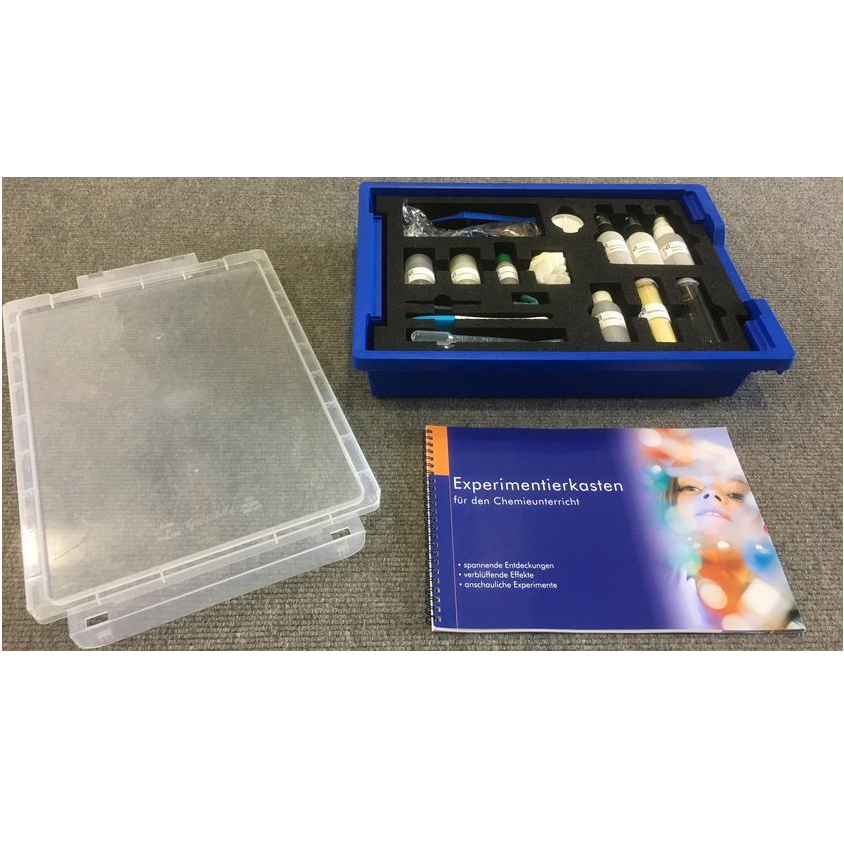
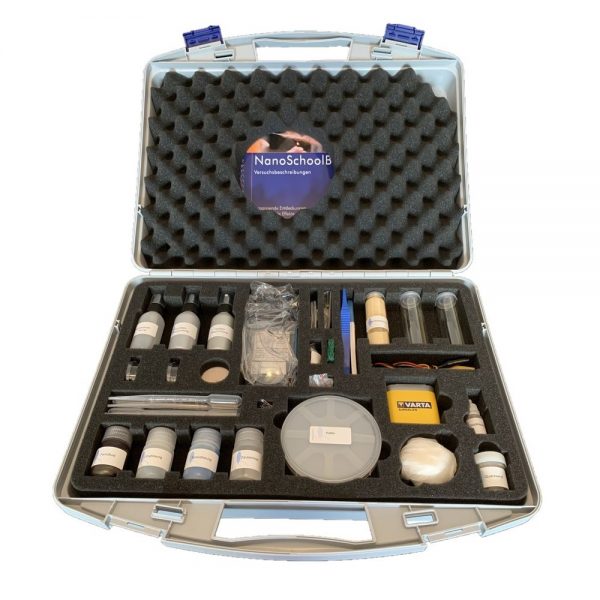
The kit includes:
1.Protective goggles
2. plastic pincers
3. candle
4. wooden flooring uncoated
5. wooden flooring coated
6. textile coating
7. fire protection
8. citrate solution
9. tenside solution
10. wood/stone coating
11. auric chloride solution
12. protective gloves
13. sheet of glass
14. abbrasive sponge
15. pregnancy test
16. pipettes
17. invisible ink
18. lycopodium spores
19. snap cap glass container
20. paint brush
Detailed experiments:
Experiment 1: Experiments to achieve the “natural cleansing” of the lotus?
The aim of this experiment is to determine whether certain materials have a hydrophobic surface. Hydrophilic fibers on some of the surface of the leaves exert adhesive forces on the water, which increase the larger their surface area (or the harder the surface of the paper). For this reason, the water drop spreads on the surface to a greater or lesser degree. In nature, this natural cleansing effect is responsible for the surface of the leaf that remains mostly clean.
Experiment 2: Hydrophobic artificial surfaces
Along with the natural lotus phenomenon found in the plant and animal kingdom, one can apply a kind of “lotus effect” to almost any type of artificial surface, ie a water repellent, hydrophobic effect. This should not always be based on a microstructure, as the hydrophobic properties can be added to surfaces by applying a coating containing fluorinated compounds, a Teflon-like effect. It should be noted, however, that a real lotus surface is always based on microstructures. Only the visible phenomenon is the same.
Experiment 3: Production of hydrophobic surface on wood or mineral material
This experiment involves the development of artificial hydrophobic surfaces in materials such as wood or stone (you will do hydrophobic [water repellent] treatment.
Experiment 4: Production of hydrophobic surface on fabrics
As in the previous experiment, you will develop a hydrophobic effect in this experiment as well. It is based on the same principle as Experiment 2. However, in this case you are dealing with a special impregnation to obtain a hydrophobic and oil-repellent (oil repellent) for fabrics and paper.


Experiment 5: Invisible ink for glass – production of hydrophilic surface (anti-fog)
Now you will learn how to develop the opposite of the hydrophobic effect, ie the hydrophilic (water-friendly) effect. What is so special about a surface that has been treated with the so-called “anti-fog coating” is the fact that it does not absorb water but that the aqueous solutions are “spread” (ie dispersed). Instead of seeing a drop, we see nothing. In this experiment we process a glass surface with a hydrophilic agent to write a hidden message on it.
Experiment 6: Scratch-resistant wood coating. In general, MDF panels (medium density fiberboard) are used to produce interior doors, front and rear pieces of furniture with wooden designs. Designs are printed using a variety of printing techniques, such as inkjet printing on panels. One problem that is often associated with this process is that the surface is particularly sensitive to mechanical stress and can therefore be easily scratched.In order to protect the designs on the MDF panels, a transparent, thin 10 mm protective coating was applied, in which nanoparticles were incorporated. This gives the panels more stability. Even if we scratch the surface with a wire brush, the panel will not scratch.

Experiment 7: Fire protection
Paper, cardboard or wood are mostly made up of cellulose, a carbohydrate. When the solution is poured onto the paper and heated, the phosphorite is chemically combined with the carbohydrate and converted to carbon, but without burning in the process. Phosphoric acid esters are formed, which decompose under heat to become phosphoric acid and carbon. Nitrogen compounds in this case have the following function:
heat causes the formation of nitrogen gas (which in itself is non-flammable) and displaces the oxygen required at the source of the fire (there is no flame without oxygen).
Experiment 8: Recognition of colloids through the Tyndall phenomenon
Using fairly simple methods you will see how the nanoclimate, colloidal systems in liquids can be detected through the Tyndall effect.
Experiment 9: Gold nanoparticle production
In the following experiment we will produce clusters of golden nanoscale that will be easily recognizable through the manifestation of a typical red ruby color.
One way to produce gold nanoparticles is through the citrate method. This method involves the production of colloidal gold or gold clusters in a solution.
The experiment is based on an oxidation-reduction reaction of tetrachloroacrylic acid (known as tetrachloroacetic acid (III) trihydrate) in which gold ions are reduced to clusters of gold atoms. Sodium citrate reducer not only serves to reduce gold, but also acts as a diluent to stabilize the created gold clusters. With the addition of the reducer, the coagulation of the metal ions stops. The result is a colloidal complex enclosed by a binder.
Experiment 10: Spitting fire from small particles. The lycopodium (also known as the wolf foot or fox tail) is a plant belonging to the genus Cryptogamous. In the past, lycopodium seeds (seeds from lycopodium clavatum) were used for medicinal purposes to dry wounds (which is why it also bears the name of witch’s flour).Today, lycopodium seeds are used in pyrotechnic products and are used in the criminal science to detect fingerprints (in combination with other substances).
Experiment 11: Hyperhydrophobia
A surface is considered hydrophobic when the contact angle between the water droplets and the surface exceeds 90 degrees. A super-hydrophobic surface is one in which the contact angle exceeds 150 degrees. A surface becomes more hydrophobic as it becomes
rougher. Such surfaces are also known as freckles surfaces, as the drop is like a freckle on a nail bed, which comes in contact with the surface only in certain places. Air pockets are formed between the contact points on these surfaces, which further reduces adhesion.


 Botzees
Botzees Keyestudio
Keyestudio Fischertechnik
Fischertechnik