Guaiacol 100g
Guaiacol 100g
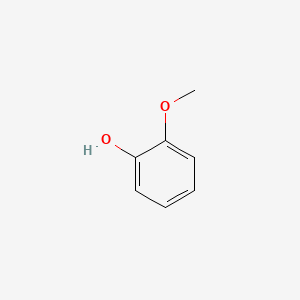
Guaiacol is an organic compound with the formula C6H4(OH)(OCH3). It is a phenolic compound containing a methoxy functional group. Guaiacol appears as a viscous colorless oil, although aged or impure samples are often yellowish. It occurs widely in nature and is a common product of the pyrolysis of wood. Guaiacol is usually derived from guaiacum or wood creosote. It is produced by a variety of plants. It is also found in essential oils from celery seeds, tobacco leaves, orange leaves, and lemon peels. The pure substance is colorless, but samples become yellow upon exposure to air and light. The compound is present in wood smoke, resulting from the pyrolysis of lignin. The compound contributes to the flavor of many substances such as whiskey and roasted coffee.
History and preparation
The compound was first isolated by Otto Unverdorben in 1826. Guaiacol is produced by methylation of o–catechol, for example using potash and dimethyl sulfate:
- C6H4(OH)2 + (CH3O)2SO2 → C6H4(OH)(OCH3) + HO(CH3O)SO2
Guaiacol can be prepared by diverse routes in the laboratory. o-Anisidine, derived in two steps from anisole, can be hydrolyzed via its diazonium derivative. Guaiacol can be synthesized by the dimethylation of catechol followed by selective mono-demethylation.
- C6H4(OCH3)2 + C2H5SNa[10] → C6H4(OCH3)(ONa) + C2H5SCH3
CAS Number: 90-05-1
IUPAC name: 2-Methoxyphenol
Properties
| C7H8O2 | |
| Molar mass | 124.139 g·mol−1 |
| Appearance | colorless oil or crystalline solid |
| Density | 1.112 g/cm3, liquid 1.129 g/cm3, crystals |
| Melting point | 26–29 °C (79–84 °F; 299–302 K) |
| Boiling point | 204–206 °C (399–403 °F; 477–479 K) |
| 23.3 g/L at 25 °C |
Structure
Safety

Guaiacol cause irritation. Skin and eye contact should be avoided.


 Botzees
Botzees Keyestudio
Keyestudio Fischertechnik
Fischertechnik