Hydrazine Hydrate 500mL
Hydrazine Hydrate 500mL
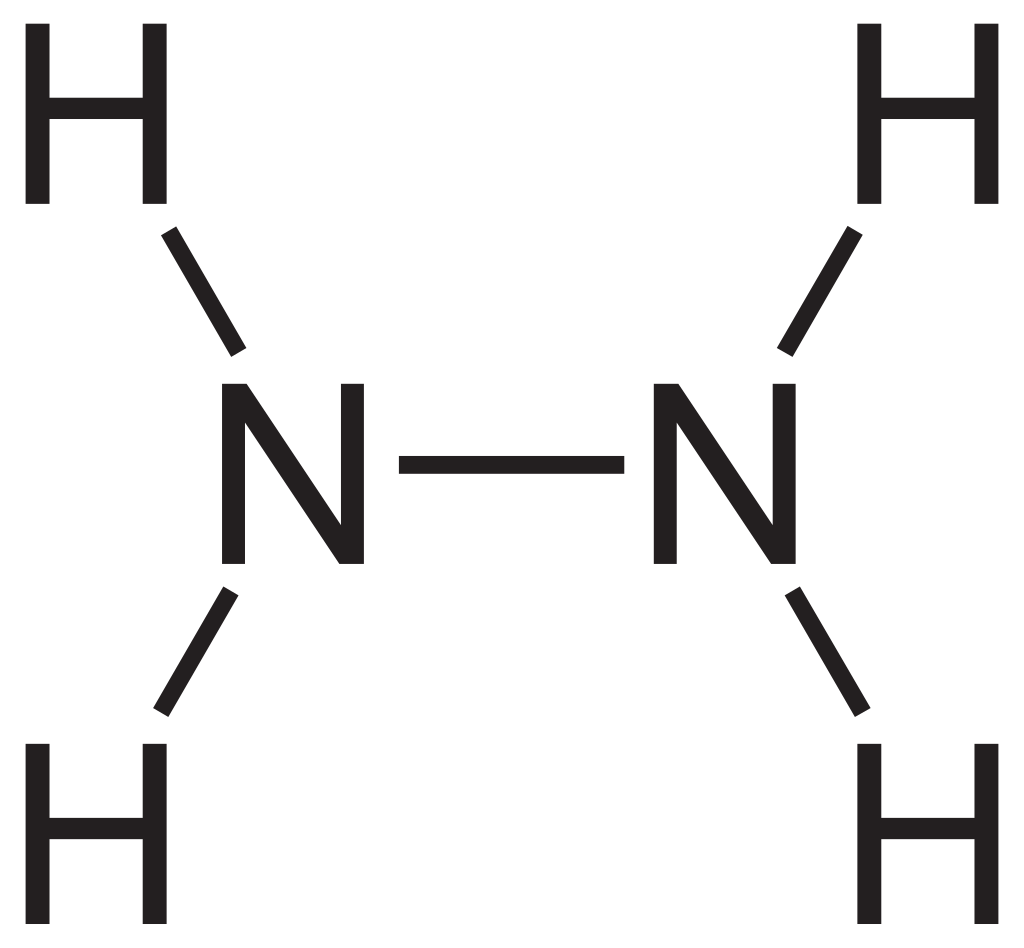
Hydrazine is an inorganic compound with the chemical formula N2H4. It is a simple pnictogen hydride, and is a colourless flammable liquid with an ammonia-like odour.
Hydrazine is highly toxic unless handled in solution as e.g., hydrazine hydrate (NH2NH2 · xH2O). As of 2015, the world hydrazine hydrate market amounted to $350 million.[8] Hydrazine is mainly used as a foaming agent in preparing polymer foams, but applications also include its uses as a precursor to polymerization catalysts, pharmaceuticals, and agrochemicals, as well as a long-term storable propellant for in-space spacecraft propulsion.
About two million tons of hydrazine hydrate were used in foam blowing agents in 2015. Additionally, hydrazine is used in various rocket fuels and to prepare the gas precursors used in air bags. Hydrazine is used within both nuclear and conventional electrical power plant steam cycles as an oxygen scavenger to control concentrations of dissolved oxygen in an effort to reduce corrosion.[9]
Hydrazines refer to a class of organic substances derived by replacing one or more hydrogen atoms in hydrazine by an organic group
Chemical Properties
| Properties | |
|---|---|
| N2H4 | |
| Molar mass | 32.0452 g·mol−1 |
| Appearance | Colorless, fuming, oily liquid[3] |
| Odor | Ammonia-like[3] |
| Density | 1.021 g·cm−3 |
| Melting point | 2 °C; 35 °F; 275 K |
| Boiling point | 114 °C; 237 °F; 387 K |
| Miscible[3] | |
| log P | 0.67 |
| Vapor pressure | 1 kPa (at 30.7 °C) |
| Acidity (pKa) | 8.10 (N2H5+)[4] |
| Basicity (pKb) | 5.90 |
| Conjugate acid | Hydrazinium |
Refractive index (nD) | 1.46044 (at 22 °C) |
| Viscosity | 0.876 cP |
Chemical Structure Hydrazine Hydrate 500mL
Safety and storage





Description
Gas producers and propellants
The majority use of hydrazine is as a precursor to blowing agents. Specific compounds include azodicarbonamide and azobisisobutyronitrile, which produce 100–200 mL of gas per gram of precursor. In a related application, sodium azide, the gas-forming agent in air bags, is produced from hydrazine by reaction with sodium nitrite.[10]
Hydrazine is also used as a long-term storable propellant on board space vehicles, such as the Dawn mission to Ceres and Vesta, and to both reduce the concentration of dissolved oxygen in and control pH of water used in large industrial boilers. The F-16 fighter jet, Eurofighter Typhoon, Space Shuttle, and U-2 spy plane use hydrazine to fuel their auxiliary power units.[11]
Precursor to pesticides and pharmaceuticals
Hydrazine compounds can be effective as active ingredients in admixture with or in combination with other agricultural chemicals such as insecticides, miticides, nematicides, fungicides, antiviral agents, attractants, herbicides or plant growth regulators.[13]
Small-scale, niche, and research
The Italian catalyst manufacturer Acta (chemical company) has proposed using hydrazine as an alternative to hydrogen in fuel cells. The chief benefit of using hydrazine is that it can produce over 200 mW/cm2 more than a similar hydrogen cell without the need to use expensive platinum catalysts.[14] As the fuel is liquid at room temperature, it can be handled and stored more easily than hydrogen. By storing the hydrazine in a tank full of a double-bonded carbon–oxygen carbonyl, the fuel reacts and forms a safe solid called hydrazone. By then flushing the tank with warm water, the liquid hydrazine hydrate is released. Hydrazine has a higher electromotive force of 1.56 V compared to 1.23 V for hydrogen. Hydrazine breaks down in the cell to form nitrogen and hydrogen which bonds with oxygen, releasing water.[14] Hydrazine was used in fuel cells manufactured by Allis-Chalmers Corp., including some that provided electric power in space satellites in the 1960s.
A mixture of 63% hydrazine, 32% hydrazine nitrate and 5% water is a standard propellant for experimental bulk-loaded liquid propellant artillery. The propellant mixture above is one of the most predictable and stable, with a flat pressure profile during firing. Misfires are usually caused by inadequate ignition. The movement of the shell after a mis-ignition causes a large bubble with a larger ignition surface area, and the greater rate of gas production causes very high pressure, sometimes including catastrophic tube failures (i.e. explosions).[15] From January–June 1991, the U.S. Army Research Laboratory conducted a review of early bulk-loaded liquid propellant gun programs for possible relevance to the electrothermal chemical propulsion program.[15]


 Botzees
Botzees Keyestudio
Keyestudio Fischertechnik
Fischertechnik