Sodium Sulfate, anhydrous 1Kg 99%
Sodium Sulfate (also known as sodium sulphate or sulfate of soda) is the inorganic compound with formula Na2SO4 as well as several related hydrates. All forms are white solids that are highly soluble in water. With an annual production of 6 million tonnes, the decahydrate is a major commodity chemical product. It is mainly used for the manufacture of detergents and in the kraft process of paper pulping.
Properties
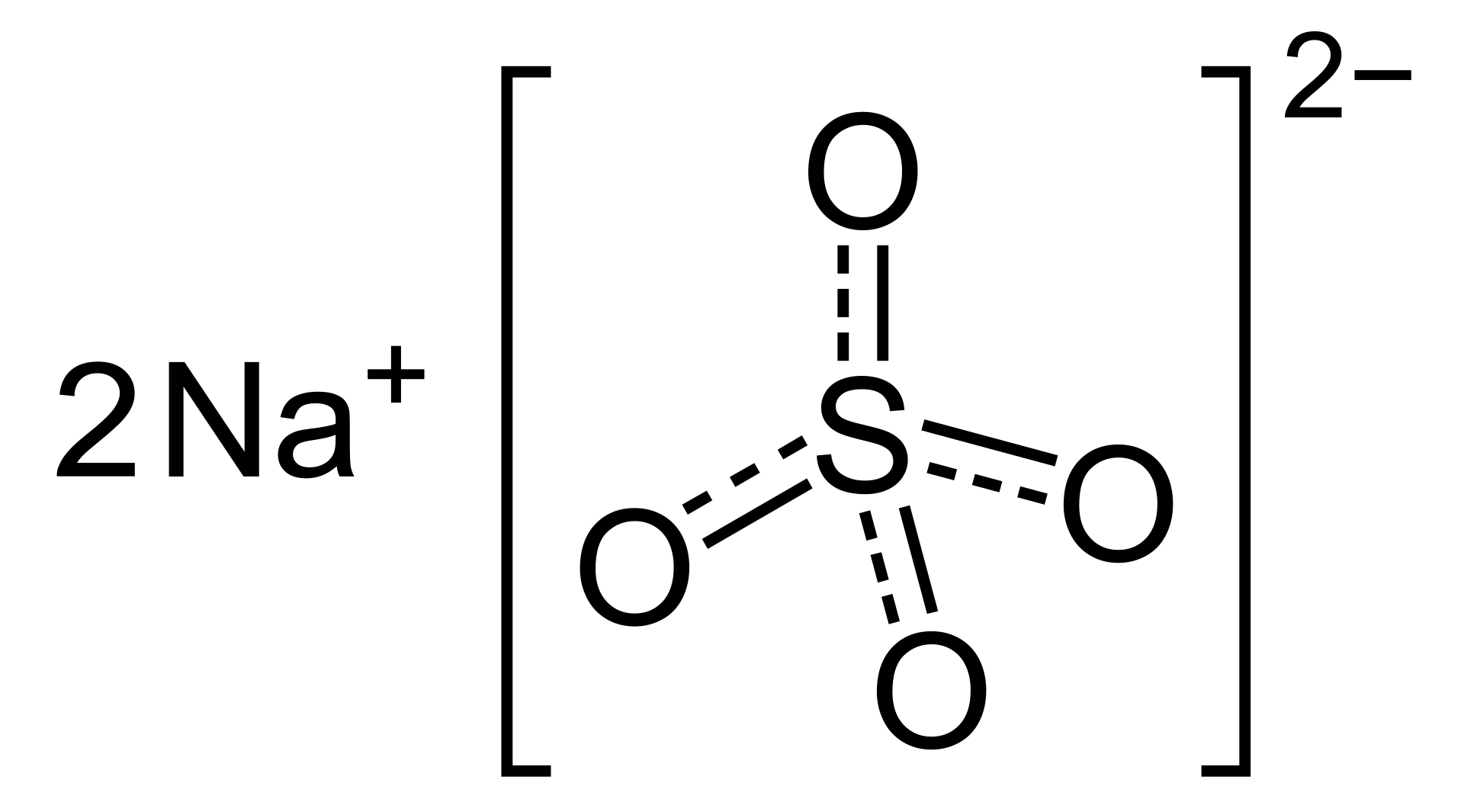
Chemical Formula: Na2SO4
Molecular weight: 142.04 g/mol (anhydrous)
Density: 2.664 g/cm3 (anhydrous)
Melting point: 884 °C (1,623 °F; 1,157 K) (anhydrous)
Boiling point: 1,429 °C (2,604 °F; 1,702 K) (anhydrous)
solubility in water: anhydrous:
4.76 g/100 mL (0 °C)
13.9 g/100 mL (20 °C)
42.7 g/100 mL (100 °C)
Chemical structure
Safety


Corrosive Irritant
Although sodium sulfate is generally regarded as non-toxic, it should be handled with care. The dust can cause temporary asthma or eye irritation; this risk can be prevented by using eye protection and a paper mask. Transport is not limited, and no Risk Phrase or Safety Phrase applies.
Description
Crystals of the decahydrate consist of [Na(OH2)6]+ ions with octahedral molecular geometry. These octahedra share edges such that eight of the 10 water molecules are bound to sodium and two others are interstitial, being hydrogen bonded to sulfate. These cations are linked to the sulfate anions via hydrogen bonds. The Na-O distances are about 240 pm. Crystalline sodium sulfate decahydrate is also unusual among hydrated salts in having a measurable residual entropy (entropy at absolute zero) of 6.32 J·K−1·mol−1. This is ascribed to its ability to distribute water much more rapidly compared to most hydrates.


 Botzees
Botzees Keyestudio
Keyestudio Fischertechnik
Fischertechnik


