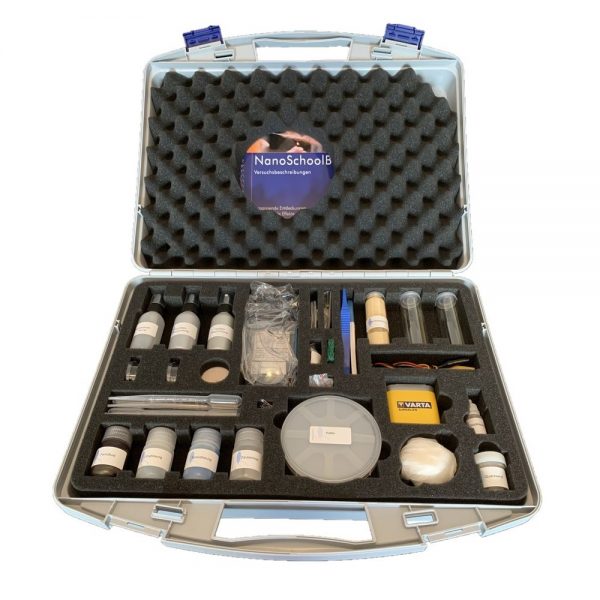
Nano experiment kit for physics
How do you get from simple quartz sand to a high-performance wafer and how does the memory effect in shape memory alloys work? Why can lycopodium be super hydrophobic and cause huge explosions at the same time? Or how do smart surface coatings work, e.g. in the automotive industry (flip-flop effect) or in the electronics industry (ITO coatings)?
This experiment kit provides relevant answers to all those questions. We hope that the experiments described here will help you pass a few interesting hours in the world of small particles and fine structures. Some of the experiments introduced here are intended for demonstration only, but others can be incorporated into lessons under the supervision of a teacher without excessive preparation.
The kit includes:
1.Protective gloves
2. Silica sand
3. plastic pincers
4. silicon waffer
5. Ferrofluid
6. tenside solution
7. rock crystal
8. petri dish
9. memory wire
10. LED
11. Protective goggles
12. batteries
13. aligator cables
14. ΙΤΟ sheet of glass
15. untreated sheet of glass
16. magnet
17. Hemisphere (effect Flip flop)
18. Lycopodium spores
19. snap cap glass container
20. pipettes
Detailed experiments:
Experiment 1: Increase electrical conductivity through the cathode (ITO) oxide
Common glass is not conductive, ie it cannot transmit electric currents. However, the special glass in this box can do this because the glass is covered with an extremely thin, invisible nanostrate, the so-called ITO layer. The term ITO means Indian-Cash Oxide, a compound of mixed Indian oxide and tin oxide. It is semi-conductive and transparent, these properties form the basis for the production of electrically conductive glass.
Experiment 2: The magnetic fieldWith the help of iron fillers, it is very easy to prove the magnetic field around a magnet, the field that pulls from the north to the south pole of a magnet and that forms closed circuits. The irons are aligned according to the power lines and try to follow them.
Experiment 3: density separation with ferrofluid. In this experiment, a non-magnetic coin, which first sinks to the bottom of the liquid due to its density, “swims” to the top in the presence of a magnetic field.
Experiment 4: Memory metal – movements of atoms in the nanostructure. This experiment involves NITINOL wire. NITINOL is the name given to almost all the stoichiometric compositions consisting of nickel and titanium that display the so-called memory effect. The wire used in this experiment has pseudo-elastic behavior.
Experiment 5: Spitting Fire from small particles. The lycopodium (also known by the common name wolf foot or fox tail) is a plant belonging to the genus Cryptogamous. In the past, lycopodium seeds (seeds from lycopodium clavatum) were used for medicinal purposes to dry wounds (which is why it is also called the witch’s flour flour).Today, lycopodium seeds are used in pyrotechnic products and are used in forensic science to detect fingerprints (in combination with other substances).
Experiment 6: Hyper Hydrophobia. A surface is considered hydrophobic when the contact angle between the water droplets and the surface exceeds 90 degrees. A super-hydrophobic surface is one in which the contact angle exceeds 150 degrees. A surface becomes more hydrophobic as it becomesrougher. Such surfaces are also known as freckles surfaces, as the drop is like a freckle on a nail bed, which comes in contact with the surface only in certain places. Air pockets are formed between the contact points on these surfaces, which further reduces adhesion.


 Botzees
Botzees Keyestudio
Keyestudio Fischertechnik
Fischertechnik