Sodium lauryl sulfate (SLS) 99% 100g
Sodium lauryl sulfate (SLS) 99% 100g
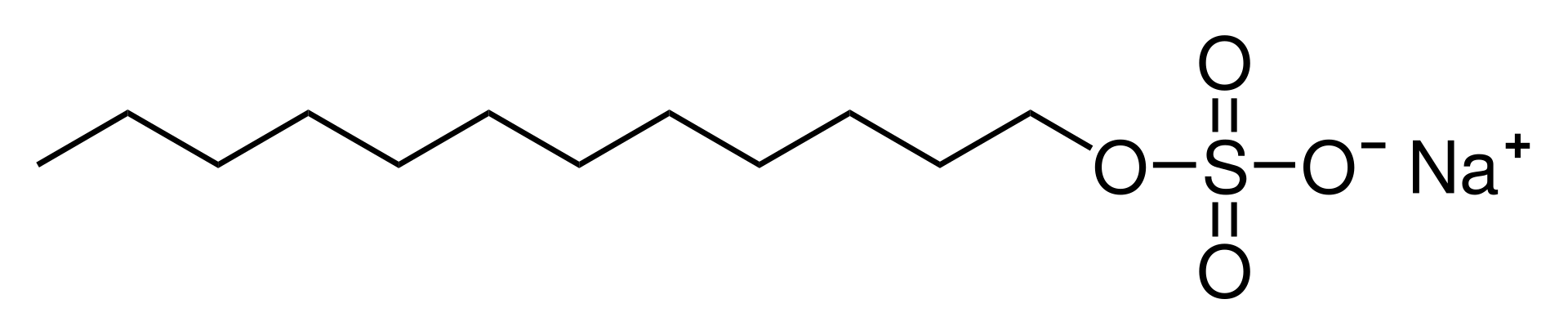
Sodium dodecyl sulfate (SDS) or sodium lauryl sulfate (SLS), sometimes written sodium laurilsulfate, is an organic compound with the formula CH3(CH2)11OSO3Na and structure H3C−(CH2)11−O−S(=O)2−O−Na+. It is an anionic surfactant used in many cleaning and hygiene products. This compound is the sodium salt of the 12-carbon organosulfate. Its hydrocarbon tail combined with a polar “headgroup” give the compound amphiphilic properties that make it useful as a detergent. SDS is also component of mixtures produced from inexpensive coconut and palm oils. SDS is a common component of many domestic cleaning, personal hygiene and cosmetic, pharmaceutical, and food products, as well as of industrial and commercial cleaning and product formulations.
Chemical Properties
| Properties | |
|---|---|
| C12H25NaSO4 | |
| Molar mass | 288.372 g/mol |
| Appearance | white or cream-colored solid |
| Odor | odorless |
| Density | 1.01 g/cm3 |
| Melting point | 206 °C (403 °F; 479 K) |
| Surface tension: | |
| 8.2 mM at 25 °C[1] | |
Refractive index (nD) | 1.461 |
Chemical Sructure Sodium laureth sulfate 1L

| Names | |
|---|---|
| Preferred IUPAC name Sodium dodecyl sulfate | |
| Other names Sodium monododecyl sulfate; Sodium lauryl sulfate; Sodium monolauryl sulfate; Sodium dodecanesulfate; dodecyl alcohol, hydrogen sulfate, sodium salt; n-dodecyl sulfate sodium; Sulfuric acid monododecyl ester sodium salt |
Safety
SDS is not carcinogenic in low concentrations according to some[clarification needed] studies. Like all detergents, sodium lauryl sulfate removes oils from the skin, and can cause skin and eye irritation. It has been shown to irritate the skin of the face, with prolonged and constant exposure (more than an hour) in young adults. SDS may worsen skin problems in individuals with chronic skin hypersensitivity, with some people being affected more than others.
Description
Production
SLS is synthesized by treating lauryl alcohol with sulfur trioxide, oleic or chlorosulfuric acid to produce hydrogen lauryl sulfate. Lauryl alcohol can be used in pure form or as a mixture of fatty alcohols. When produced from these sources, “SDS” products are a mixture of sodium alkyl sulfates with SDS as the primary component.
SLS is commercially available in powder, pellet, other forms (differing in dissolution rates), as well as in aqueous solutions of various concentrations.
Uses
Cleanliness and hygiene
SLS is mainly used in laundry detergents with many cleaning applications. It is a highly effective surfactant and is used in any job that requires the removal of greasy stains and residues. For example, it is found in higher concentrations with industrial products including engine degreasers, floor cleaners and car cleaners.
It is an ingredient in hand soap, toothpastes, shampoos, shaving creams and shower gels due to its foaming ability, surfactant properties and thickening action.
Laboratory applications
SDS is used in purification procedures and is commonly used as a component to lyse cells during RNA extraction or DNA extraction and to denature proteins in preparation for electrophoresis in the SDS-PAGE technique.
Protein denaturation using SDS
In the case of SDS-PAGE, the compound works by breaking the non-covalent bonds in the proteins and thus denatures them. By binding to proteins at a ratio of one SDS molecule per 2 amino acid residues, the negatively charged detergent provides all proteins with a similar net negative charge and therefore a similar charge-to-mass ratio. Thus, the difference in the mobility of the polypeptide chains in the gel is attributed solely to their length and not so much to their physical charge as to their shape. This separation based on the size of the polypeptide chain simplifies the analysis of protein molecules.
Pharmaceutical applications
SLS is used in the pharmaceutical sector as an ionic solubilizer and emulsifier suitable for applications in liquid dispersions, solutions, emulsions and microemulsions, tablets, foams and semi-solids such as creams, lotions and gels. In addition, SLS aids tablet wettability as well as lubrication during manufacture. Brand names of pharmaceutical grade SLS include Kolliphor SLS and Kolliphor SLS Fine.
Different applications
SLS is used in an improved technique for preparing brain tissues for light microscopy study. The technique, which has been named CLARITY, was the work of Karl Deisseroth and his collaborators at Stanford University and involves infusing the organ with an acrylamide drug to bind the organ’s macromolecules (proteins, nucleic acids, etc.); by thermal polymerization to form a “brain-hydrogel” and then by removal of lipids using SDS to examine light scattering with min. loss of protein, making it almost transparent.


 Botzees
Botzees Keyestudio
Keyestudio Fischertechnik
Fischertechnik




